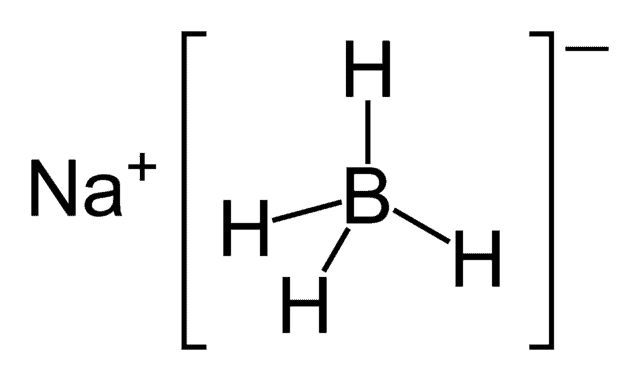
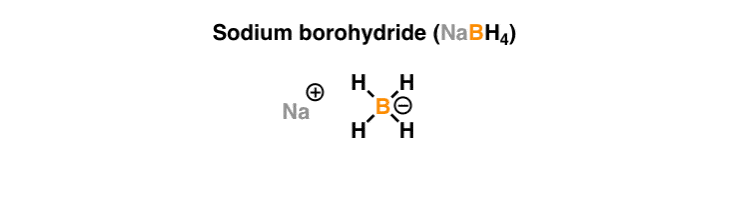
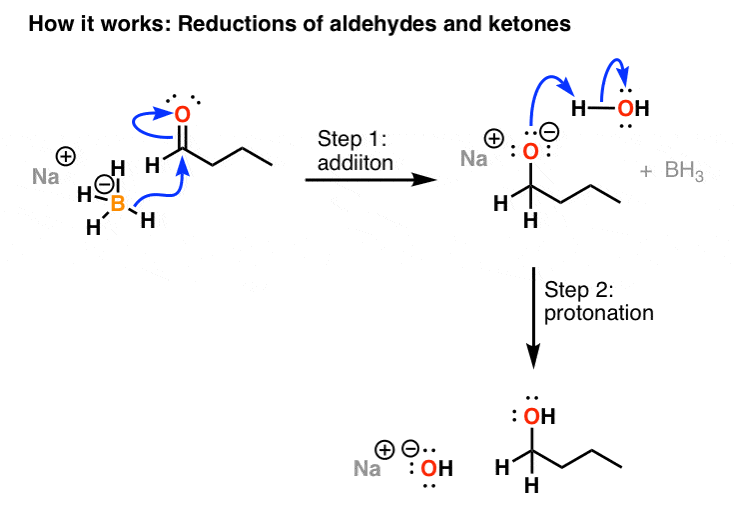
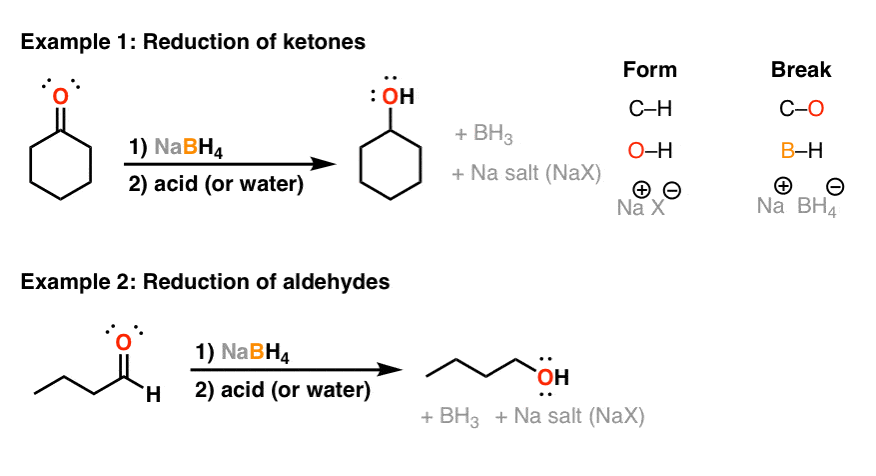
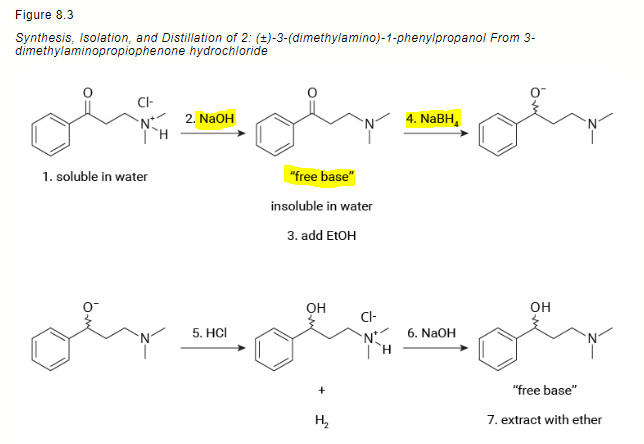
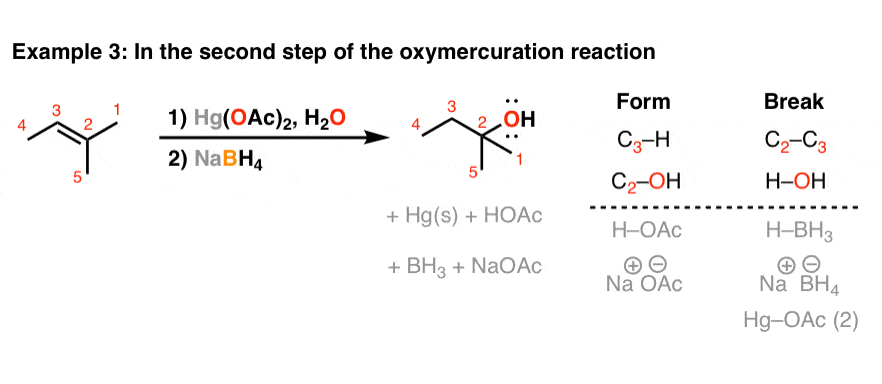
Reaction of InCl3 with Various Reducing Agents: InCl3–NaBH4-Mediated Reduction of Aromatic and Aliphatic Nitriles to Primary Amines | The Journal of Organic Chemistry
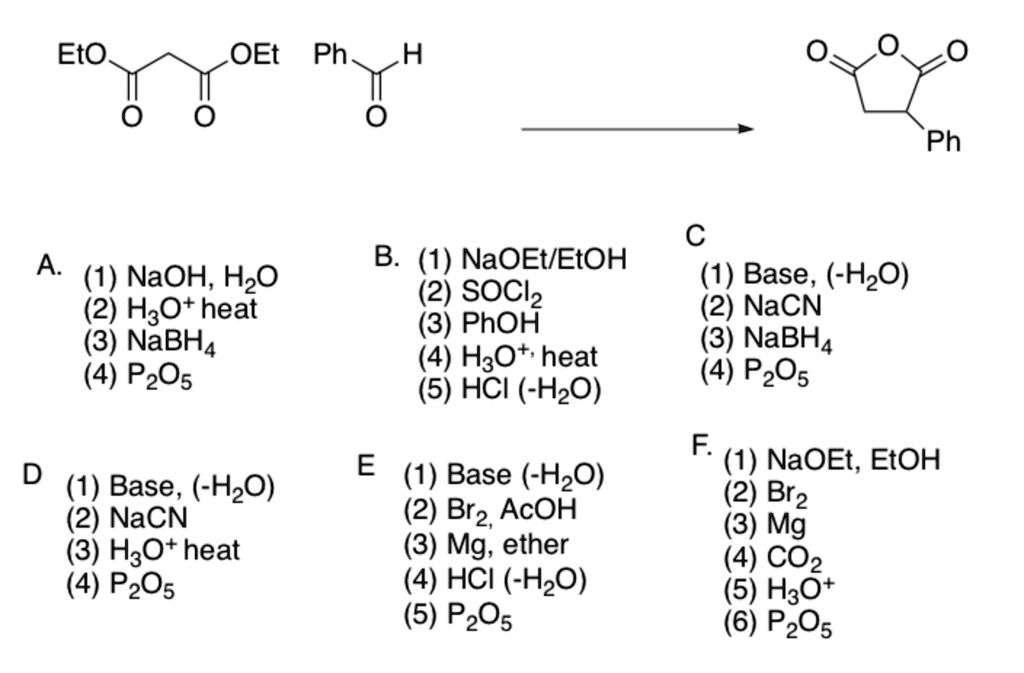
SOLVED: Eto OEt Ph H Ph C (1) Base, (-HzO) (2) NaCN (3) NaBH4 (4) PzO5 A NaOH, HzO (2) H3Ot heat (3) NaBH4 (4) PzOs B: (1) NaOEtIEtOH (2) SOClz 3)